Before 2020, very few people were concerned about research on contagious viruses and vaccine development. Of course that all changed after COVID-19. One topic in particular which has gained in popularity over the last few years is Biosafety Level 4 Labs. Take a look at the Google Trends below:
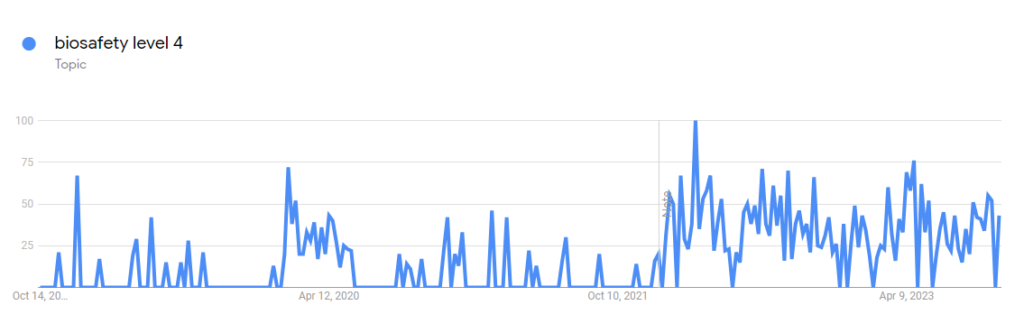
You can see a jump in interest during 2020 and another jump in 2022. These events corollate with the start of the Pandemic in 2020 and the increased discussion around a possible lab leak in 2022. Due to this increased interest in the topic we have decided to answer the question, What Viruses Are Considered Biosafety Level 4 (BSL-4)?
Characteristics Of Biosafety Level 4 Viruses
Biosafety Level 4 (BSL-4) is the highest level of containment for laboratories working with infectious agents. BSL-4 laboratories are designed to handle the most dangerous and exotic pathogens, including viruses, which pose a high risk to both humans and the environment.
Here are some characteristics of viruses and agents that are typically handled in BSL-4 laboratories:

- Highly Pathogenic: BSL-4 viruses are characterized by their extreme pathogenicity. They can cause severe and often fatal diseases in humans, and there are usually no vaccines or effective treatments available.
- No Known Cure or Vaccine: Many BSL-4 viruses have no known cure or vaccine, making them particularly dangerous. Examples include the Ebola virus, Marburg virus, and certain strains of the Lassa virus.
- Strict Containment: BSL-4 laboratories have strict containment measures in place to prevent accidental exposure and release of these pathogens. These measures include specialized air filtration systems, positive pressure suits (space suits), and double-sealed doors to prevent any escape of the virus.
- Stringent Protocols: Researchers working with BSL-4 viruses must adhere to stringent safety protocols and are often required to undergo extensive training. All procedures must be meticulously planned and executed to minimize the risk of exposure.
- Limited Access: Access to BSL-4 laboratories is highly restricted, and entry is typically limited to trained and authorized personnel. Visitors are subject to strict controls and must adhere to safety protocols.
- Research and Vaccine Development: BSL-4 laboratories are used for researching these highly dangerous pathogens to better understand their biology, transmission, and potential treatments or vaccines. This research is critical for public health preparedness.
Examples of BSL-4 Viruses
- Ebola Virus: The Ebola virus causes Ebola virus disease, which is a severe and often fatal illness in humans. There are several species of Ebola virus, and some have caused outbreaks with high mortality rates in Africa.
- Marburg Virus: The Marburg virus is related to Ebola and causes Marburg virus disease, which is similarly severe and often fatal.
- Lassa Virus (Certain Strains): While not all strains of Lassa virus require BSL-4 containment, some highly pathogenic strains do. Lassa fever can cause severe illness in humans and has a high mortality rate.
- Crimean-Congo Hemorrhagic Fever Virus: This virus causes Crimean-Congo hemorrhagic fever, a severe illness that can result in bleeding and organ failure.
- Nipah Virus: The Nipah virus can cause severe respiratory and neurological disease in humans. It is known to be transmitted from bats to humans and has caused outbreaks in Asia.
Cleaning And Decontaminating A Biohazard Level 4 (BSL-4) Laboratory
Due to the extremely dangerous pathogens, cleaning and decontamination procedures are essential for BSL-4 Laboratories. Here are some general steps and considerations for cleaning a BSL-4 laboratory:
- Personal Protective Equipment (PPE): All personnel involved in the cleaning and decontamination process must wear appropriate personal protective equipment (PPE), including positive pressure suits, gloves, masks, and other necessary gear to prevent any contact with the pathogens.
- Decontamination Chambers: BSL-4 laboratories often have decontamination chambers, which are used to safely enter and exit the laboratory. These chambers help prevent the escape of pathogens. Personnel will undergo decontamination procedures upon exiting the laboratory.
- Isolation and Segregation: The laboratory should be isolated from other areas of the facility to prevent contamination of non-BSL-4 areas. Separate ventilation systems are often used to maintain negative pressure within the BSL-4 laboratory, ensuring that air does not flow out of the laboratory.
- Disinfectants: High-level disinfectants and chemical agents are used to decontaminate surfaces and equipment within the laboratory. The choice of disinfectant may depend on the specific pathogen, and it should be effective against the agent being handled.
- Autoclaving: Autoclaving is a common method for sterilizing laboratory equipment and waste. BSL-4 laboratories typically have autoclaves designed for the inactivation of highly pathogenic agents.
- Decontamination Procedures: The laboratory should have established decontamination procedures, which include a sequence of steps for cleaning and decontaminating surfaces and equipment. These procedures must be rigorously followed and may involve the use of chemical disinfectants, steam sterilization, or other methods, depending on the situation.
- Waste Management: Waste generated in the BSL-4 laboratory should be treated as highly infectious and hazardous. Proper disposal methods, such as incineration, should be used to ensure complete inactivation of the pathogens.
- Documentation: Detailed records of cleaning and decontamination procedures should be maintained, including the date and time of cleaning, the disinfectants used, and the personnel involved.
- Quality Assurance: BSL-4 laboratories typically have quality assurance programs to ensure that cleaning and decontamination procedures are followed correctly and effectively. These programs may involve regular audits and inspections.
- Decontamination Validation: After cleaning and decontamination, laboratory surfaces and equipment should be tested to ensure that no viable pathogens are present. This may involve surface sampling and culture to confirm that decontamination has been successful.
It’s important to note that only trained and authorized personnel should be involved in the cleaning and decontamination of BSL-4 laboratories. The procedures are highly regulated and designed to minimize the risk of accidental exposure and release of dangerous pathogens.
Contact Bio Recovery Today
BSL-4 laboratories play a crucial role in understanding and combating these highly dangerous viruses, but their use is strictly regulated to ensure safety and prevent accidental releases. At Bio Recovery we provide state of the art remediation services and have many years of experience dealing with infectious diseases.
Laboratories of all kind can rely on us for regular decontamination and waste disposal to ensure you are taking the right steps to keep your facility safe. Give us a call at (888) 664-2599
